What is proteomics?
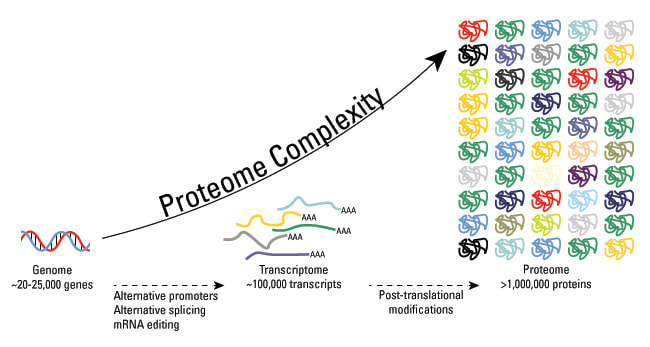
The large scale study of proteins within an organism is considered proteomics. It is an emerging and necessary field of study outside of transcriptomics as proteins are complex and dynamic due to their constant alterations that take place after translation. Scientists are coming to the realization that the final product of a gene is inherently more complex and closer to function than the gene itself. Additionally, post translational modifications alter proteins so in order to understand the complete function of proteins they must be studied on the proteomic level [1].
Proteomics have become possible to study through the improvement of protein databases and mass spectrometry, the primary method for studying proteins.
Proteomics have become possible to study through the improvement of protein databases and mass spectrometry, the primary method for studying proteins.
Sub-cellular location of SYNGAP1
SYNGAP1 is a non-secreted protein found in the cytoplasm and nucleus region of a cell. It is trafficked back and forth as it is largely a regulatory protein. The majority of proteins SYNGAP1 interacts with are also found in these locations.
What are post-translational modifications?
|
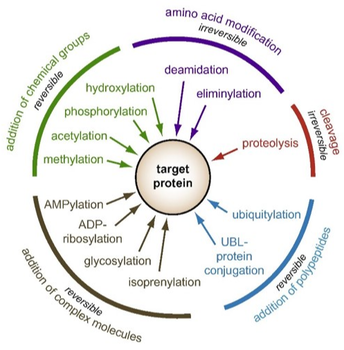
Post-translational modifications are a set of diverse biological processes that chemically alter the amino acids within a protein after it has been translated by the ribosome [2]. Some common examples include the addition of chemical groups such as phosphates (phosphorylation) or ubiquitin (ubiquitination), proteolytic cleavage, or addition of bonds like disulfide bridges [3]. These modifications can either be made permanent or may change over time. In general, post-translational modifications increase the functional diversity of the proteome and can alter an individual proteins stability, folding, targeting or recognition [4].
|
Protein Phosphorylation
|
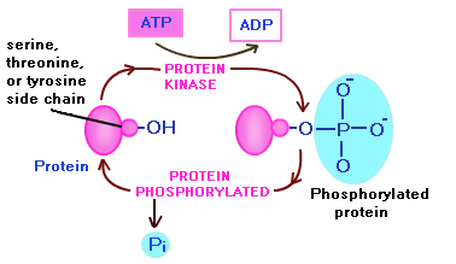
Phosphorylation is one of the most common post-translational modifications and is simply the action of kinases adding phosphate groups onto amino acids within the protein. Often times threonine, serine and tyrosine are the phosphorylated amino acids. Phosphorylation allows for a major molecular mechanism through which protein function can be regulated in response to extracellular stimuli. Virtually all types of extracellular signals produce most of their diverse physiological effects by regulating phosphorylation of specific phosphoproteins in their target cells. The majority of neuronal proteins are regulated by phosphorylation [5].
|
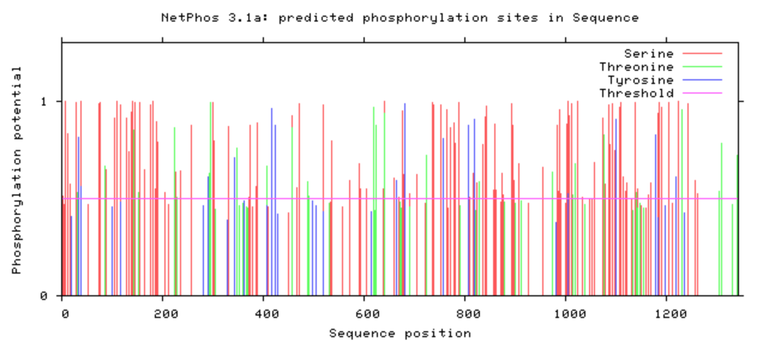
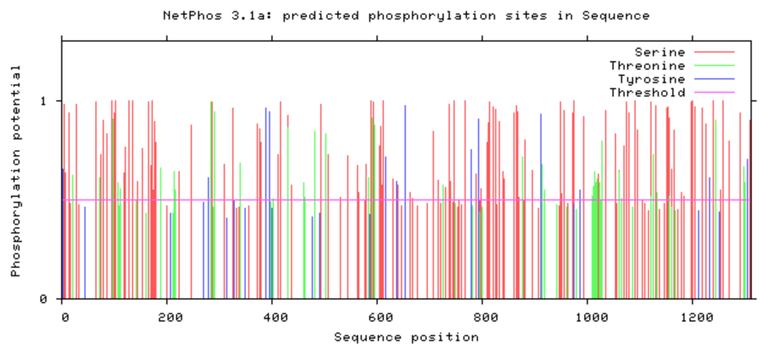
SYNGAP1 Phosphorylation Sites
Human SYNGAP1
Zebrafish SYNGAP
Discussion
As shown above SYNGAP1 carries a large amount of phosphorylation sites and a decent proportion of these seem to be conserved between humans and zebrafish, but there are also some points of difference that would be worth further researching. Phosphorylation sites would be an interesting aspect to study surrounding SYNGAP1 considering it's vast roles and functions within neurons. It is highly probable that these sites greatly range depending on the proteins function at that particular moment and could cause mutations if affected.
References
[1] Graves, Paul R, and Timothy A J Haystead. “Molecular biologist's guide to proteomics.” Microbiology and molecular biology reviews : MMBR vol. 66,1. (2002).
[2] https://www.sciencedirect.com/topics/neuroscience/posttranslational-modification
[3] https://www.khanacademy.org/science/biology/gene-regulation/gene-regulation-in-eukaryotes/a/regulation-after-transcription
[4] https://www.thermofisher.com/us/en/home/life-science/protein-biology/protein-biology-learning-center/protein-biology-resource-library/pierce-protein-methods/overview-post-translational-modification.html
[5] Nestler Eric, Greengard Paul. "Protein Phosphorylation is of Fundamental Importance in Biological Regulation." NCBI. Basic Neurochemistry: Molecular, Cellular and Medical Aspects. 6. 1999
Images:
Header: https://biologywise.com/cell-nucleus-structure-functions
Figure 1: https://www.thermofisher.com/us/en/home/life-science/protein-biology/protein-biology-learning-center/protein-biology-resource-library/pierce-protein-methods/overview-post-translational-modification.html
Figure 2: http://ptm-biosciences.com/15/protein-modifications
Figure 3: http://www.writeopinions.com/phosphorylation
Figure 4 and 5: Generated by Abby Jaquish using NetPhos
[2] https://www.sciencedirect.com/topics/neuroscience/posttranslational-modification
[3] https://www.khanacademy.org/science/biology/gene-regulation/gene-regulation-in-eukaryotes/a/regulation-after-transcription
[4] https://www.thermofisher.com/us/en/home/life-science/protein-biology/protein-biology-learning-center/protein-biology-resource-library/pierce-protein-methods/overview-post-translational-modification.html
[5] Nestler Eric, Greengard Paul. "Protein Phosphorylation is of Fundamental Importance in Biological Regulation." NCBI. Basic Neurochemistry: Molecular, Cellular and Medical Aspects. 6. 1999
Images:
Header: https://biologywise.com/cell-nucleus-structure-functions
Figure 1: https://www.thermofisher.com/us/en/home/life-science/protein-biology/protein-biology-learning-center/protein-biology-resource-library/pierce-protein-methods/overview-post-translational-modification.html
Figure 2: http://ptm-biosciences.com/15/protein-modifications
Figure 3: http://www.writeopinions.com/phosphorylation
Figure 4 and 5: Generated by Abby Jaquish using NetPhos
This webpage was produced as an assignment for Genetics 564, an undergraduate capstone at University of Wisconsin- Madison.